
Table of Contents
Intro
Motor vehicles are a significant source of air pollution because the combustion process in the engine does not yield complete elements. Nitrogen combines with oxygen to produce nitrogen oxide which is harmful to the environment. For instance, it creates an ozone gas to form that inhibits plant growth. Furthermore, carbon monoxide and hydrocarbons are other by-products of the combustion process that produce problems for the ecosystem. This write-up explores the topic of catalytic converters in detail and provides you with as much information you need to understand the role they play in sustainability and in your motor vehicle.
What are Catalytic Converters
Catalytic converters are a device used to control the three major pollutants that exit from the exhaust of gasoline-powered vehicles. In 1975 and 1976, the EPA created new mandates for emissions levels to be lower than what the previous technology was already doing. As a result, manufacturers created the catalytic converter that is installed on most 1975 and new auto vehicles. There are no separate parts that make up the catalytic converter it is simply an all in one device that is very sturdy and difficult to damage.
Each catalytic converter needs an AIR injection system to protect it. The AIR injection system helps the catalytic converter by moving the air that would otherwise enter the exhaust manifold into the atmosphere. Essentially, it prevents damage from overheating.
More technology is used in protecting the expensive piece of equipment that the catalytic converter is. For example, parts that make up a catalyst protection system include a vacuum differential, by-pass valve, manifold vacuum, temperature switch, and a solenoid.
The Pollutants
There are three main pollutants that come out of a car or truck’s exhaust pipe. They are:
Nitrogen oxide
Oxides of nitrogen are combining with hydrocarbons to form photochemical smog. This smog when in interaction with the sun creates an ozone gas in the troposphere or at the ground level.
Cat converter nitros oxide
Nitrogen oxide is a generic classification for primarily two types of gases mainly nitric oxide and nitrogen dioxide. These are common air pollutants that contribute to photochemical smog, acid rain, and ozone gases in the troposphere. Fuel is nitrogen bound, and when the combustion occurs at high temperatures, it causes the nitrogen atoms to turn into free radicals. Eventually, to stabilize themselves, the nitrogen atoms combine with oxygen and form oxides of nitrogen.
Oxides of nitrogen can occur with volatile organic compounds to form toxic by-products such as nitroarenes, nitrate radical, and nitrosamines. These toxic substrates can lead to DNA mutations.
Nature does have its way of removing oxides of nitrogen from the air through hydroxyl radical reactions that form nitric acid. Nitric acid can be eliminated through geological deposition.
Diesel is a liquid which is made from fractionally distilling petroleum fuel oil. It is the process of distillation that makes diesel a distinct fuel type. T here are sub-types of diesel fuel such as biodiesel, BTL, GTL but petrol-diesel which is standardized as ultra-low sulfur diesel is the most common version. However, diesel fuel contributes significantly to higher amounts of nitrogen oxides because the combustion temperature is higher in the vehicle’s engine. Most trucks that impose heavy loads on the drive cycle and engine contribute to higher combustion temperatures resulting in evermore NOx.
Hydrocarbons
Hydrocarbons are one of the leading sources of fuel in the world. However, anthropogenic hydrocarbon production has led to widespread contamination of soil which leads to deterioration in plant life and adversely affects human health too.
Cat converter hydrocarbons
There are two primary reasons why hydrocarbons cannot combust completely; engine misfire and quench. Engine misfire happens when no hydrocarbons are burned. Quench areas from where the flame goes out in the combustion chamber before the hydrocarbons have completely burned. Quench areas exist in several places. For instance, the area where the head gasket protects the cylinder head to the block is noted as a quench area. The area between the first compression ring and the top piston is also a quench area.
Carbon monoxide
Carbon monoxide is another pollutant that is released through the vehicle exhaust. Carbon monoxide can cause poisoning which may result in headaches, mental fatigue, and even death – given there is enough content of it in the air. Carbon monoxide also contributes to photochemical smog formation. Carbon monoxide forms when there is not enough air in the combustion chamber to burn the fuel completely. As a result, carbon monoxide is fractionally burned fuel. Carbon monoxide is made up of 1 carbon atom and one oxygen atom which are intertwined through a triple bond of two covalent and dative bonds. Motor vehicles are not the only source of urban carbon monoxide pollution, but CO produces itself through lawn mower, power washers, back-up generators, and burning of fossil fuels such as natural gas/coal.
How Does a CAT Converter Control Emissions?
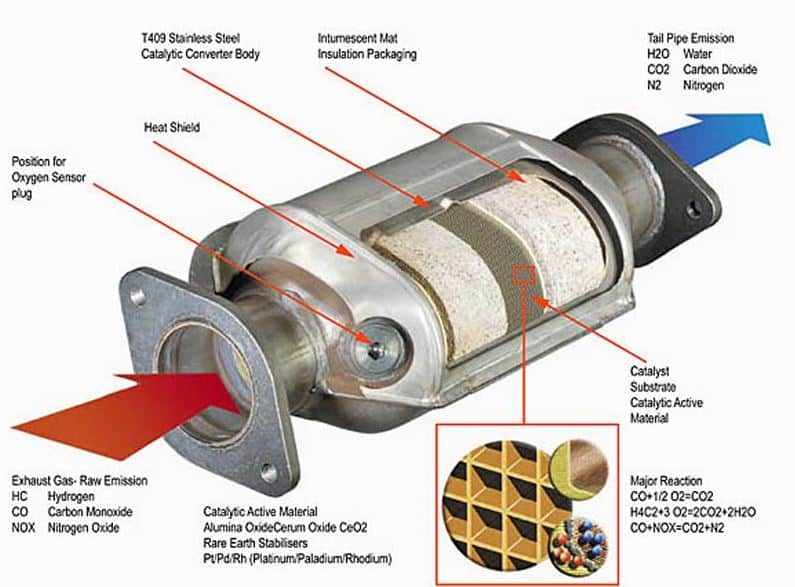
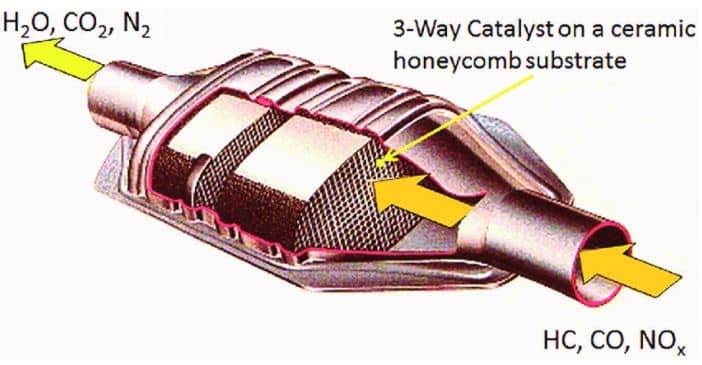
Catalytic converters work to control air pollution of the three main contaminants which are nitrogen oxide, hydrocarbons, and carbon monoxide. It works much like a muffler system because it is built into the same drive shaft. The converter is normally positioned in the exhaust system close to the manifold. If your car has a dual exhaust system, it will also have two catalytic converters. The outer shell of the CAT is made of rugged stainless steel. The internal designs vary between monolith and pellet.
A monolith design includes a flow diffuser, stainless steel mesh, and honeycomb substrate. The flow diffuser works to spread the exhaust gases more consistently through the monolith. The purpose of the mesh is to protect the ceramic material from road impact. The monolith honeycomb substrate contains pallidum, platinum, rhodium, and ceramic material.
A pellet type catalytic converter contains internal components such as baffles, plates, insulation, and aluminum oxide among its outer stainless-steel shell. The purpose of the baffle is to direct the exhaust gas up and through the aluminum oxide pellets. It is a cheaper type of catalytic converter and not as durable as the monolith.
Catalyst Reactions
The primary metals that catalyze the contaminating pollutants are pallidum, platinum, and rhodium. These precious metals create catalysts which speed up the chemical reactions needed to convert the substances into water vapor and carbon dioxide when they leave the exhaust. The method in which the CAT uses to oxidize hydrocarbons, nitrogen oxides, and carbon monoxide is through burning at high temperatures. Normal catalytic converters operate at 1400 degrees Fahrenheit.
When there is more gasoline in the fuel mixture than air, it is called running rich as opposed to running lean. Rich mixtures often caused by a spark plug misfire increase the temperature of the converter. If the temperature reaches 2500 degrees Fahrenheit, the aluminum oxide pellets in the pellet type converter will be damaged. Luckily, you can repair a pellet catalytic converter, but it is rarely done so. Therefore, it is important to check if any spark plug wires are open or disconnected in order to avoid a CAT failure.
Final words
In conclusion, catalytic converters are a durable automobile component that plays an integral role in oxidizing the free radicals created during the combustion process of the vehicle’s engine. There are precious metals used to initiate the oxidation of the free radicals and mechanisms in place to protect the very integrity of the entire system. If you adhere to good maintenance practices and do your part in achieving a sustainable transportation system for all, then you will enjoy the benefits that the catalytic converter has to offer for you and your vehicle.
 by
by